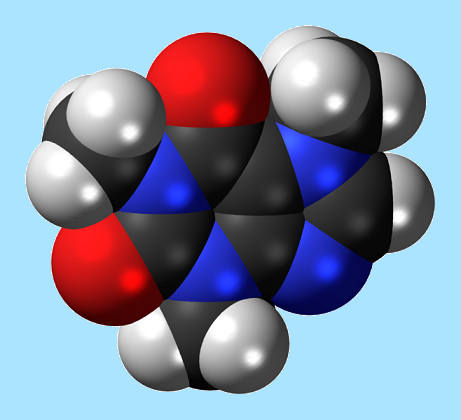
Caffeine Molecule
Caffeine is a bitter, white crystalline xanthine alkaloid and a stimulant drug. Caffeine is found in varying quantities in the seeds, leaves, and fruit of some plants, where it acts as a natural pesticide that paralyzes and kills certain insects feeding on the plants, as well as enhancing the reward memory of pollinators. It is most commonly consumed by humans in infusions extracted from the seed of the coffee plant and the leaves of the tea bush, as well as from various foods and drinks containing products derived from the kola nut. Other sources include yerba maté, guarana berries, guayusa, and the yaupon holly.
In humans, caffeine acts as a central nervous system stimulant, temporarily warding off drowsiness and restoring alertness. It is the world's most widely consumed psychoactive drug, but unlike many other psychoactive substances, it is legal and unregulated in nearly all parts of the world. Beverages containing caffeine, such as coffee, tea, soft drinks, and energy drinks, enjoy great popularity. In North America, 90% of adults consume caffeine daily.
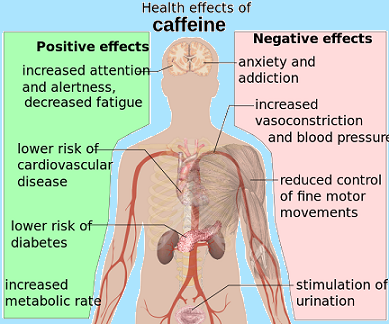
Part of the reason caffeine is classified by the Food and Drug Administration as GRAS (generally recognized as safe) is that toxic doses (over 10 grams) are much higher than typically used doses (less than 500 milligrams). Ordinary consumption can have low health risks, even when carried on for years there may be a modest protective effect against some diseases, including Parkinsons Disease, and certain types of cancer. Caffeine can have both positive and negative effects on anxiety disorders. Some people experience sleep disruption if they consume caffeine, especially during the evening hours, but others show little disturbance and the effect of caffeine on sleep is highly variable. Evidence of a risk to pregnancy is equivocal, but some authorities have concluded that prudent advice is for pregnant women to limit consumption to the equivalent of two cups of coffee per day or less. The American Congress of Obstetricians and Gynecologists (ACOG) concluded in 2010 that caffeine consumption is safe up to 200 mg per day in pregnant women.
Caffeine has pressor and mild diuretic effects when administered to people who are not used to it, but regular users develop a tolerance to this effect, and studies have generally failed to support the common notion that ordinary consumption contributes significantly to dehydration. With heavy use, tolerance develops rapidly to autonomic effects such as elevated heart rate and muscle twitching, but not to the cognitive or arousal effects of caffeine. The degree to which caffeine can produce clinically significant physical and mental dependence remains a subject of controversy in the medical literature.
Health effects
Stimulant effects
Caffeine is a central nervous system and metabolic stimulant, and is used both recreationally and medically to reduce physical fatigue and to restore alertness when drowsiness occurs. It produces increased wakefulness, faster and clearer flow of thought, increased focus, and better general body coordination. The amount of caffeine needed to produce effects varies from person to person, depending on body size and degree of tolerance. Effects begin less than an hour after consumption, and a moderate dose usually wears off in about five hours. Caffeine has a number of effects on sleep, but does not affect all people in the same way. It improves performance during sleep deprivation but may lead to subsequent insomnia. In shift workers it leads to fewer mistakes caused by tiredness.In athletics, moderate doses of caffeine can improve sprint, endurance, and team sports performance, but the improvements are usually not very large. Interestingly, some evidence suggests that coffee does not produce the ergogenic effects observed in other caffeine sources. High doses of caffeine, however, can impair athletic performance by interfering with coordination. Evidence shows that, contrary to common advice, caffeine may be helpful at high altitude.
Physical effects
Consumption of large amounts of caffeine usually more than 250 mg per day can lead to a condition known as caffeinism.Caffeinism usually combines caffeine dependency with a wide range of unpleasant physical and mental conditions including nervousness, irritability, restlessness, insomnia, headaches, and heart palpitations after caffeine use.
Psychological effects
Four caffeine-induced disorders are recognized by the American Psychiatric Association (APA) including: caffeine intoxication, caffeine-induced sleep disorder, caffeine-induced anxiety disorder and caffeine-related disorder not otherwise specified (NOS). The DSM-IV defines caffeine-induced sleep disorder, as an individual who regularly ingests high doses of caffeine sufficient to induce a significant disturbance in his or her sleep, sufficiently severe to warrant clinical attention. As of 2010 the effect of caffeine on people withADHD is not known. However anecdotal evidence suggests that many individuals with ADHD already use caffeine to self-medicate themselves or their dependants, and they find that it has the opposite effect than it normally does, such as inducing a calm-down effect that encourages sleep instead of making them more active and stimulated. Some studies have however found a modest protective against Alzheimer disease, but the evidence is inconclusive. Caffeine can have both negative and positive effects on anxiety disorders. A number of clinical studies have shown a positive association between caffeine andanxiogenic effects and/or panic disorder. At high doses, typically greater than 300 mg, caffeine can both cause and worsen anxiety or, rarely, trigger mania orpsychosis. In moderate doses caffeine may reduce symptoms of depression and lower suicide risk. In moderate doses caffeine typically does not affect learning or memory,and can improve cognitive functions, especially in people who are fatigued, possibly due to its effect on alertness. However anxiety sufferers can have high caffeine sensitivity. For some people, anxiety can be very much reduced by discontinuing caffeine use.
Chemical properties and biosynthesis
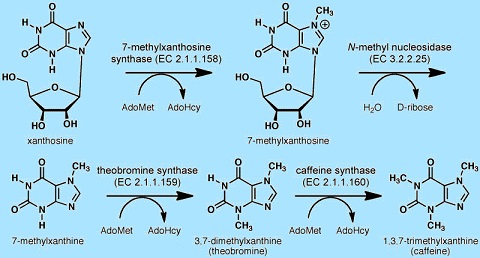
The two amide groups of caffeine exist predominately as zwitterionic resonance structures where the nitrogen and carbon atoms are double bonded to each other so that both of these nitrogen atoms are essentially planar (in sp2 orbital hybridization). The fused ring system therefore contains a total of ten pi electrons and hence according to Hückel's rule isaromatic. Caffeine is synthesized in plants from the purine nucleotides AMP, GMP, and IMP. These in turn are transformed intoxanthosine and then theobromine, the latter being the penultimate precursor of caffeine. Being readily available as a byproduct of decaffeination, caffeine is not usually synthesized chemically. If desired, it may be synthesized from dimethylurea and malonic acid. Pure anhydrous caffeine is a white colorless powder with a melting point of 227228 °C. Caffeine is moderately soluble in water at room temperature (2 g/100 mL), but very soluble in boiling water (66 g/100 mL). It is also moderately soluble in ethanol (1.5 g/100 mL). It is weakly basic (pKa = ~0.6) requiring strong acid to protonate it.
Pharmacology
Inside the body caffeine acts through several mechanisms, but its most important effect is to counteract a substance calledadenosine that naturally circulates at high levels throughout the body, and especially in the nervous system. In the brain, adenosine plays a generally protective role, part of which is to reduce neural activity levels for example, there is some evidence that adenosine helps to induce torpor in animals that seasonally hibernate.
Mechanism of action
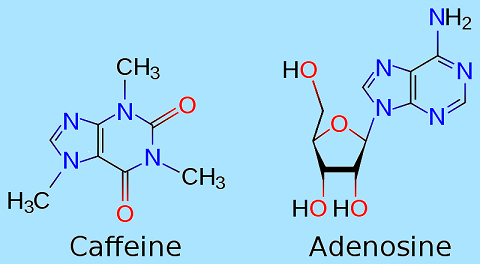
Caffeine's primary mechanism of action is as anantagonist of adenosine receptors in the brain Adenosine acts as an inhibitor neurotransmitter that suppresses activity in the central nervous system. Consumption of caffeine antagonizes adenosine and increases activity in neurotransmission including acetylcholine, epinephrine, dopamine, serotonin,glutamate, norepinephrine, cortisol, and in higher doses, endorphins which explains the analgesic effect to some users. At very high doses (exceeding 500 milligrams) caffeine inhibits GABA neurotransmission. This evidence explains why caffeine causes anxiety, insomnia, rapid heart and respiration rate. Because caffeine is both water-soluble and lipid-soluble, it readily crosses the bloodbrain barrier that separates the bloodstream from the interior of the brain. Once in the brain, the principal mode of action is as a nonselective antagonist of adenosine receptors(in other words, an agent that reduces the effects of adenosine). The caffeine molecule is structurally similar to adenosine, and is capable of binding to adenosine receptors on the surface of cells without activating them, thereby acting as a competitive inhibitor. Adenosine is found in every part of the body, because it plays a role in the fundamental adenosine triphosphate (ATP) related energy producing mechanism and is also needed for RNA synthesis, but it has additional functions in the brain. The evidence indicates that brain adenosine acts to protect the brain by suppressing neural activity and by increasing blood flow via receptors located on vascular smooth muscle. Brain adenosine levels are increased by various types of metabolic stress, including lack of oxygen and interruption of blood flow. There is evidence that adenosine functions as a synaptically released neurotransmitter in some parts of the brain; however, stress-related adenosine increases appear to be produced mainly by extracellular metabolism of ATP. Unlike most neurotransmitters, adenosine does not seem to be packaged into vesicles that are released in a voltage-controlled manner, but the possibility of such a mechanism has not been ruled out fully. Several classes of adenosine receptors have been described, with different anatomical distributions. A1 receptors are widely distributed, and act to inhibit calcium uptake. A2Areceptors are heavily concentrated in the basal ganglia, an area that plays a critical role in behavior control, but can be found in other parts of the brain as well, in lower densities. There is evidence that A 2A receptors interact with the dopamine system, which is involved in reward and arousal. (A2A receptors can also be found on arterial walls and blood cell membranes.) Beyond its general neuroprotective effects, there are reasons to believe that adenosine may be more specifically involved in control of the sleep-wake cycle. Robert McCarley and his colleagues have argued that accumulation of adenosine may be a primary cause of the sensation of sleepiness that follows prolonged mental activity, and that the effects may be mediated both by inhibition of wake-promoting neurons via A1 receptors, and activation of sleep-promoting neurons via indirect effects on A2A receptors. More recent studies have provided additional evidence for the importance of A2A, but not A1, receptors. Caffeine, like other xanthines, also acts as a phosphodiesterase inhibitor. A number of potential mechanisms have been proposed for the athletic performance-enhancing effects of caffeine. In the classic, or metabolic theory, caffeine may increase fat utilization and decrease glycogen utilization. Caffeine mobilizes free fatty acids from fat and/or intramuscular triglycerides by increasing circulating epinephrine levels. The increased availability of free fatty acids increases fat oxidation and spares muscle glycogen, thereby enhancing endurance performance. In the nervous system, caffeine may reduce the perception of effort by lowering the neuron activation threshold, making it easier to recruit the muscles for exercise.
Caffeine metabolites
Metabolites of caffeine also contribute to caffeine's effects. Paraxanthine is responsible for an increase in the lipolysis process, which releases glycerol and fatty acids into the blood to be used as a source of fuel by the muscles. Theobromine is a vasodilator that increases the amount of oxygen and nutrient flow to the brain and muscles. Theophyllineacts as a smooth muscle relaxant that chiefly affects bronchioles and acts as a chronotrope and inotrope that increases heart rate and force of contraction.
Metabolism
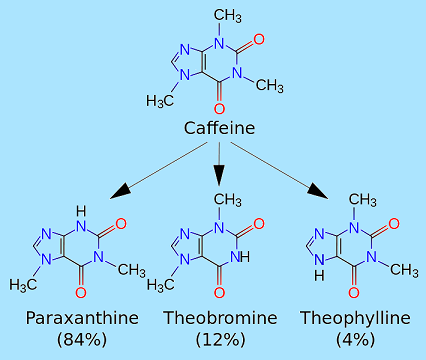
Caffeine is metabolized in the liver into three primary metabolites: paraxanthine (84%),theobromine (12%), and theophylline (4%) Caffeine from coffee or other beverages is absorbed by the small intestine within 45 minutes of ingestion and then distributed throughout all tissues of the body. Peak blood concentration is reached within one hour. It is eliminated by first-order kinetics. Caffeine can also be absorbed rectally, evidenced by the formulation of suppositories of ergotamine tartrate and caffeine (for the relief of migraine) and chlorobutanol and caffeine (for the treatment of hyperemesis). The biological half-life of caffeine the time required for the body to eliminate one-half of the total amount of caffeine varies widely among individuals according to such factors as age, liver function, pregnancy, some concurrent medications, and the level ofenzymes in the liver needed for caffeine metabolism. It can also be significantly altered by drugs or hormonal states. In healthy adults, caffeine's half-life has been measured with a range of results. Some measures get 4.9 hours, and others are at around 6 hours. Heavy cigarette smokers show a decrease in half-life of 3050%, oral contraceptives can double it, and pregnancy can raise it even more, to as much as 15 hours during the last trimester. In newborn infants the half-life can be 80 hours or more; however it drops very rapidly with age, possibly to less than the adult value by the age of 6 months. The antidepressantfluvoxamine (Luvox) reduces the clearance of caffeine by more than 90%, and prolongs its elimination half-life more than tenfold; from 4.9 hours to 56 hours. Caffeine is metabolized in the liver by the cytochrome P450 oxidase enzyme system, in particular, by the CYP1A2 isozyme, into three dimethylxanthines, each of which has its own effects on the body: Paraxanthine (84%): Increases lipolysis, leading to elevated glycerol and free fatty acid levels in the blood plasma. Theobromine (12%): Dilates blood vessels and increases urine volume. Theobromine is also the principal alkaloid in the cocoa bean, and therefore chocolate. Theophylline (4%): Relaxes smooth muscles of the bronchi, and is used to treat asthma. The therapeutic dose of theophylline, however, is many times greater than the levels attained from caffeine metabolism. Each of these metabolites is further metabolized and then excreted in the urine. Caffeine can accumulate in individuals with severe liver disease, increasing its half-life. Some quinolone antibiotics exert an inhibitory effect on CYP1A2, thereby reducing clearance of caffeine and thus increasing blood levels. A 2011 analysis published by PLoS Genetics reviewed five studies covering more than 47,000 subjects of European descent. Researchers determined that habitual caffeine intake is associated with variations in two genes that regulate how quickly the body processes caffeine. Subjects who had a high-intake mutation of either gene on both chromosomes consumed 40 mg more caffeine per day (equivalent to a can of cola) than people who did not.
My Project
Before starting the modeling phase i performed a study of the proportions using pen and paper.
The purpose of this study was to select the units of appropriate measurement to accurately reproduce the perfect misure of the molecule.
Each point of the pattern is a triple of values ??corresponding to the coordinates of the spheres centre of the 3D model (x, y ,z) .
The first step of the modeling phase was performed by reproducing a circle function and a cylinder function to represent the ties.
The following is a code fragment that represents the CIRCLE and CYLINDER functions.
var CIRCLE_PLANE = function (r) {
var mapcircle = function (v) {
alpha = v[0];
r = v[1];
return [r*COS(alpha), r*SIN(alpha)];
}
dom2D = PROD1x1([INTERVALS(2*PI)(36), INTERVALS(r)(1)]);
return MAP(mapcircle)(dom2D);
}
var CYLINDER = function (r,h) {
var surface = CIRCLE_PLANE(r);
var cylinder = EXTRUDE([h])(surface);
cylinder = COLOR([0,0,0])(R([0,2])(PI/2)(cylinder));
return cylinder;
}
Spheres that represent atoms.
The second step of the modeling phase was performed by reproducing a parametric sphere function to create a various spheres with different color and dimension. (passing only the coordinates x,y,z)
var redSphere = function(tx,ty,tz){
var domainf=DOMAIN([[0,PI],[0,2*PI]])([36,36]);
var mappingRedBlack=function(v){
var a=v[0];
var b=v[1];
var u=0.5*SIN(a)*COS(b);
var v=0.5*SIN(a)*SIN(b);
var w=0.5*COS(a);
return[u,v,w];};
var sphereA=MAP(mappingRedBlack)(domainf);
sphereA= COLOR([1,0,0])(sphereA);
sphereA = T([0,1,2])([tx,ty,tz])(sphereA);
return sphereA;
}
The third step of the modeling phase was performed by reproducing a ties for a various atoms.
This has been possible by implementing a distance function that calculates the distance between two centers, given a pair of atoms. Thereafter, for each bond, were performed 1 or 2 rotations, the first axis Z and the second on if and only if the atoms were located on different floors.
A code of distance function
var distance = function(p1,p2) {
p1x = p1[0];
p1y = p1[1];
p1z = p1[2];
p2x = p2[0];
p2y = p2[1];
p2z = p2[2];
var distance3D = Math.sqrt( Math.pow((p2x-p1x),2) + Math.pow((p2y-p1y),2) + Math.pow((p2z-p1z),2) );
return distance3D;
}
In conclusion, the bonds were combined with the atoms completing the molecular structure of caffeine.
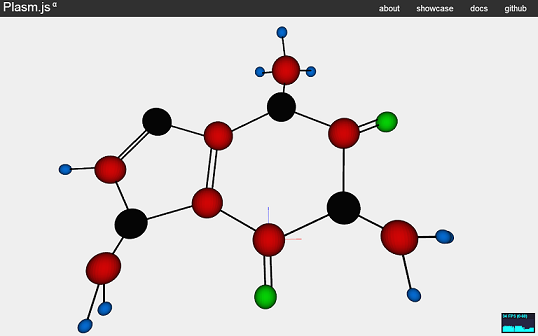
The caffeine molecule view from another angle.
